Functions of Microbial Symbionts in Fungus-farming Beetles
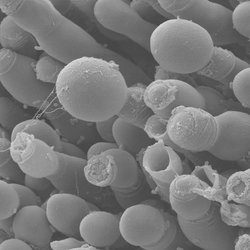

Ambrosia beetles, like the fruittree pinhole borer Xyleborinus saxesenii, live within self-dwelled systems in wood. On the walls of the tunnels and chambers, they grow their fungal gardens. The gardens are dominated by two food fungi, but other microbes grow there as well. Some mold fungi seem to be parasitic, and they negatively affect the beetles’ offspring numbers. In this project, we aim at gaining insights into the factors influencing the composition of the microbial community in the beetles’ nests and the mechanisms of beetle fungiculture.
In a first step, we identify the community of microbes associated with two native ambrosia beetle species, the fruittree pinhole borer X. saxesenii and the black timber bark beetle Xylosandrus germanus. We investigate the succession in which different microbes occur within the nests. Subsequently, we test how these microbes interact with each other. To this end, we apply state-of-the-art molecular and visualizing techniques (i.e., next-generation sequencing, SEM, FISH).
Previous studies have indicated that X. saxesenii beetles are able to influence the fungi growing in their nests in two ways: First, they are able to suppress the spread of molds. Second, they can enhance the growth and fruiting of their food fungus. Only in presence of beetles, the fungus produces fruiting bodies, so called conidia. The underlying mechanisms of these effects are also studied in this project. Preliminary results indicate that the beetles make use of other symbionts, which hence represent additional mutualistic player in this system, besides the beetles and their food fungus.
People involved in this project:
Pamela Baumann
(Bachelor and Intership projects)
Maximilian Lehenberger
(Master and PhD project)
Collaborators:
Dan Vanderpool
Microbial genomics and symbiosis, Univ. of Montana, US
Miroslav Kolarik
Institute of Microbiology, Czech Academy of Science, Prague, CZ
Social Immunity in Ambrosia Beetles & Chemical Recognition of Fungal Symbionts

In complex insect societies, the different individuals live in close spatial association and are highly related to each other. These two factors should favor the spread of parasites, but also the evolution of social behaviors to fight them, apart from chemical defense mechanisms. In the first part of this project, we study the social defense mechanisms in the ambrosia beetle Xyleborinus saxesenii. To this end, we experimentally expose them to pathogenic microorganisms, namely mold fungi. First results show that the beetles actively up-regulate hygienic behaviors like grooming each other (allo-grooming) and themselves (self-grooming), thereby likely reducing parasite load on their body surfaces, and cannibalism, thereby getting rid of infected individuals. Such collective pathogen defenses (social immunity) have been unknown in beetles so far.
In the second part of this project, we test if the beetles use olfactory cues to orient themselves during dispersal in order to locate a suitable habitat, i.e., a tree that has recently died and probably has specific chemical characteristics. In a pilot study, we found that the beetles are attracted to the volatiles of their main mutualistic fungus and to ethanol, which is emitted by recently dead trees due to degradation processes. This indicates that the beetles select new breeding grounds based on the presence of conspecifics that have already found a fungus garden successfully, and on the status of the host tree. Surprisingly, we did not find a repellent effect of volatiles produced by pathogenic fungi.
People involved in this project:
Jon Andreja Nuotclà
(MSc and PhD project)
Dep. Behavioural Ecology, University of Bern, CH
Janina Diehl
(PhD project)
Collaborators:
Prof Michael Taborsky
Dep. Behaviour Ecology, University of Bern, CH
Gerrit Holighaus,
Dep Forest Zoology, University of Göttingen, DE
Dineshkumar Kandasamy,
Dep Biochemistry, MPI-CE, DE
Prof Marko Rohlfs,
Dep. Population Ecology, University of Bremen, DE
Yeganeh Gharabigloozare,
Dep. Biochemistry, MPI-CE, DE
Further reading:
Nuotclà (2015): Master thesis.
Available from author on request.
Diversity of Fungal Symbionts of Ambrosia Beetles
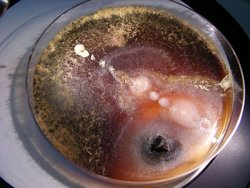
Ambrosia beetles live in tunnel systems (‘galleries’) within the wood of recently dead or dying trees, where they plant a mixture of fungi on the tunnel walls. Exact compositions of these fungal communities are hardly studied, but it is known that they are dominated by beetle-specific ambrosia fungus species. Ambrosia fungi are Ascomycetes belonging to the genera Raffaelea (Sordariomycetidae), Ambrosiella (Ceratocystidaceae) or Fusarium (Hypocreales). They produce swollen hyphens, which serve as the beetles’ sole food source. It is known that the fungi reproduce asexually, but it has never been investigated if sexual reproduction also occurs. When females leave their natal nest and disperse, they carry asexual spores to their new nest, a mechanism called vertical transmission. However, it is also not fully clear if occasional transmission between existing nests (horizontal transmission) is possible.
If the fungi only reproduce asexually, and if vertical transmission is the only way spores reach new nests, the result would be low genetic diversity of fungi among beetle populations. In this project, we determine the genetic diversity of fungi from beetle populations all around Central Europe. The aim is to unravel the respective diversity of Ambrosiella or Raffaelea genotypes across and within beetle populations. Our results will help to understand how frequently horizontal exchange of symbiotic fungi occurs between nests, and they will shed light on the occurrence and frequency of sexual reproduction of the fungi. In addition, new fungi species will be identified in collaboration with T. Harrington.
People involved in this project:
Janina Diehl
(PhD project)
Collaborators:
Prof Tom Harrington
Department of Plant Pathology, Iowa State University, US
Further reading:
Interactions of Native and Invasive Ambrosia Beetles

International movement of non-native forest insects poses a severe threat to forests worldwide. Bark and ambrosia beetles (Curculionidae: Scolytinae) represent one of the most successful groups of invaders, in part because they are easily transported in wood products and packaging material. As international trade continues to steadily increase, the risks of introducing these organisms are also on the rise. Understanding why bark and ambrosia beetles establish so easily in habitats outside their native range is recognized as a research priority by both scientists and forest managers, with the aim to promote risk assessment and management strategies for non-native species.
In this project, we want to assess what effect non-native ambrosia beetle species can have on native ones. Therefore, we test the competitive abilities of a native versus a non-native ambrosia beetle by rearing them together in artificial media. Furthermore, to investigate if competitive effects are mediated by the symbiotic fungi of the beetles, we test the main fungal associate of the two beetle species against each other on agar plates.
Principal investigator:
Davide Rassati,
DAFNAE-Entomology, University of Padova, IT
Symbionts of Parasitic Dendroctonus Bark Beetles

Bark beetles (Curculionidae: Scolytinae) of the genus Dendroctonus live in symbiosis with fungi and are widely used as model insects to study symbiosis. This is because they cover a wide range of ecological niches on the one hand, and on the other hand they have a huge economic impact. There are aggressive species that kill the trees that they inhabit, and others that inhabit stressed or dead trees. A third, parasitic group of Dendroctonus species develop in living trees without killing them. This third group is hardly studied, but very interesting as their hosts unlike dead trees are continuously defending themselves and it is unknown how the beetles overcome the tree defences.
Therefore, the beetles associate with peculiar bacterial and fungal microorganisms that help them to overcome the tree defenses. In this study, we use complementary culture-dependent and culture-independent methods to provide the first comprehensive overview of the bacterial and fungal symbionts of the parasitic beetles D. micans, D. punctatus, and D. valens. We also compare their microbial communities with those of other tree-inhabiting insects. Our first results show that many bacteria and yeasts are shared among the three species and their developmental stages, especially nitrogen-fixing and detoxifying microorganisms. Our study casts new light on the symbionts of bark beetles, and will contribute to understand how symbioses affect insects with various ecologies.
People involved in this project:
Loic Dohet
(PhD project)
Biological Control and Spatial Ecology Lab (LUBIES), Université Libre de Bruxelles, BE
Collaborators:
Jean-Claude Gregoire
Biological Control and Spatial Ecology Lab (LUBIES), Université Libre de Bruxelles, BE
Further reading:
Social Behaviour of the Coffee Berry Borer

The coffee berry borer (Hypothenemus hampei) is the major pest of coffee, causing immense yield losses in coffee-producing countries worldwide. If we want to assess potential methods for biocontrol of this species, we need to better understand its life cycle and behavior, as well as its interaction with pathogens.
The beetle spends most of its life cycle inside the coffee berry. In order to elucidate the behavior of the coffee berry borer, we developed a diet sandwich, consisting of coffee berry borer artificial diet within two glass plates. This construction allows for observing the beetles inside their nests. We are currently determining the behaviors of the larvae, males, mated and unmated females. We are particularly curious if we find signs of a social lifestyle, namely delayed dispersal of adult offspring from their mother’s nest and their engagement in cooperative tasks and breeding. Further, we test how the beetles react towards an experimental injection of microbial pathogens (e.g., Beauveria bassiana) into their artificial diet in order to explore the potential of using natural enemies for biocontrol of this pest.
Principal investigator:
Fernando Vega,
US Department of Agriculture, Maryland, US
Further reading:
Responses of Fungal Endophytes of Black Poplar to Aphids

Galling Pemphigus aphids induce major changes in the defense chemistry of black poplar. In this project we determine how these chemical changes affect the plants' fungal pathogen and endosymbiont communities.
We collect leaf samples with and without aphids in the field, determine their defense chemistry and culture fungi from these samples. In addition we check how major fungal players interact with each other and how they affect the leaf chemistry in experimental trials.
Collaborator:
Sybille Unsicker,
Dep. Biochemistry, MPI-CE
Interactions of Thistles, Endosymbiotic Fungi, and Beetle Herbivores

Like many other plants, thistles are often infected with endosymbiotic fungi that grow within their leaves. The plants are inoculated with the fungi when tortoise beetles feed on them, as they vector fungal spores.
In this project, we studied the community of endosymbiotic fungi in the cabbage thistle Cirsium oleraceum and the creeping thistle Cirsium arvense (also referred to as the Canada thistle). We were interested in wheter the beetles gain fitness benefits from feeding on fungus-infested leaves. To this end, we conducted feeding experiments using infected versus non-infected cabbage thistles and the thistle tortoise beetle Cassida rubiginosa.
People involved in this project:
Margo Wisselink,
Laboratory of Genetics, Wageningen University, NL
Collaborators:
Prof Duur Aanen
Laboratory of Genetics, Wageningen University, NL
Development of an Application Strategy for SPLAT®Verb for Population Management of European Spruce Bark Beetles (Ips typographus) with the anti-aggregation Pheromone Verbenone

The Verblps project aims to test the effectiveness of the use of SPLAT®Verb against Ips typographus aggregation and infestation. While the pheromone dispenser SPLAT®Verb is already used to manage and contain a variety of bark beetle outbreaks for species such as the Mountain pine beetle (Dendroctonus ponderosae) in the United States, its effectiveness has yet to be tested for the European spruce bark beetle Ips typographus.
SPLAT®Verb is a commercially produced and environmentally friendly pheromone dispenser that provides the controlled release of the anti-aggregation pheromone verbenone in the field. Verbenone is a naturally occurring anti-aggregation pheromone used by a variety of bark beetle species including I. typographus.
During this project, the effectiveness of the use of SPLAT®Verb against Ips typographus aggregation and infestation will be tested. The active inhibitory range, duration of effect and required quantities will be determined, its use for area protection will be investigated and application options presented.
This research is a joint project carried out by Chair of Forest Entomology and Forest Protection and the Chair of Ecosystem Physiology at the University of Freiburg together with the Forest Research Institute of Baden-Württemberg (FVA).
Funded by:
FNR - Fachagentur Nachwachsende Rohstoffe e. V.
Further reading:
Bark Beetle Sound Production: Acoustics of the European Spruce Bark Beetle Ips typographus

Passive acoustic techniques may offer a remote, non-invasive, automated alternative to traditional methods in the detection and monitoring of hard-to-access species. In recent applications, incidental sounds produced by wood borers—such as those generated in the process of feeding or moving through galleries—have been used for the early detection of some species in imported wooden packaging goods. Although a species of high economic interest, no literature exists on the incidental sounds of the European spruce bark beetle Ips typographus.
Through a series of laboratory oscilloscope measurements, we carried out an investigation into the performance of currently available acoustic equipment in detecting Ips typographus incidental sounds. Additionally, we described incidental sounds in larval, pupal and adult Ips typographus, by comparing recordings taken in infested and uninfested logs. A series of video recordings were further collected of artificial plexiglas sandwiches infested with larval, pupal and adult Ips typographus to allow for acoustic-visual correlations between the identified sounds and movement.
Our measurements revealed high disparities between the compared recordings in terms of sensitivity and signal-to-noise ratio, highlighting the importance of the two parameters when measuring a low amplitude acoustic signal such as incidental bark beetle sounds. Furthermore, we found incidental sounds of Ips typographus to be characterized by broad-band, short, irregular pulses ranging between 100Hz and 22kHz.
Our findings can be a useful point of departure for future investigations into the acoustics of Ips typographus, and may provide a stepping stone for eventual applications of acoustic technology in the early detection, monitoring and management of this species.
People involved in this project:
Funded by:
MLR BW, FVA-WS
Integrating Biodiversity to Evaluate and Optimize Timber-Harvesting Practices

The effects of timber harvesting machinery on the soil structure of forest skid trails have been relatively well investigated. Soil compaction alters soil physical parameters, leading to a decreased soil macroporosity and to an impaired pore continuity. This process sets a chain of reactions in motion, altering the gas and water balance of skid trail soils and presumably their species composition. However, the state of knowledge about the effects of heavy machinery traffic on biodiversity does not yet allow for clear interpretation and assessments. A combined analysis of soil physical, soil ecological and entomological investigations is missing, as is the determination of the site characteristics which mediate the effects of traffic on soil organisms. The goal of our study is to develop an evaluation guide for forest soil compaction based on these interrelationships, with microbial, soil faunistic and entomological examinations at the core of this development work. Thus, the open question that we aim to address is how the degree of soil compaction affects the biodiversity of soil organisms, exploring the link between the abiotic indicators of the disturbance intensity and the susceptibility of the biotic community to this influence.
Collaborators:
Prof Friederike Lang, Chair of Soil Ecology, University of Freiburg
Helmer Schack-Kirchner, Chair of Soil Ecology, University of Freiburg
Kenton Stutz, Chair of Soil Ecology, University of Freiburg
Funded by:
BfN - Bundesamt für Naturschutz
Food quality of various broadleaved trees for maturation feeding of adult cockchafer (Melolontha hipposcastani)

In the Upper Rhine region, climate change poses massive challenges for forestry. In addition to
environmental changes due to climate change such as drought and heat, the local cockchafer
populations represent another dimension. The potential changes in the tree species spectrum will be
assessed in terms of risk from cockchafer and incorporated into a Species Distribution Modeling
(SDM) as scenarios. This arises because of differences in food quality for females during their
maturation feeding on leaves of deciduous trees, which affects egg production and thus reproductive
potential. Here, the differences between native and new alternative tree species, which are
considered to be climate stable, will be experimentally elaborated in two replicates. In addition to
the established tree species red oak, sessile oak and littleleaf linden, downy oak, Norway maple and
the neophytic black cherry will be tested in the feeding experiments.
People involved in this project:
Collaborators:
in cooperation with FVA, Abteilung Waldschutz
Understanding the role of symbionts during beetle development
Beetles as holometabolous organisms go through dramatic developmental changes during their life. They are hatching from an egg laid inside a tree, developing through a larval phase adapted to feed, going through intense body modification during pupation, to continue developing into an adult which will distribute and found new nests.
We need to understand which microorganisms are involved in the different life stages, where they come from (are they environmentally derived or vertically passed from generation to generation) and what they do in the system. We will try to tackle this by using a combination of sequencing (who is there and how does the community change under certain conditions?), cultivation (having them “in our hands” will give us options to further characterize them) and experiments under controlled lab conditions as well as under natural conditions.
This fundamental approach links closely to the other research foci in our research group.
People involved in this project:
Vienna Kowallik
Study systems:
In particular but not limited to: Xyleborinus saxesenii, Xylosandrus germanus
Microbiome and Sociality
In weevils, including bark beetles, we find a huge range of social systems ranging from merely solitary (plant-feeding weevils) to aggregation (some bark and ambrosia beetles), to parental care (all bark and ambrosia beetles) to even alloparental care and reproductive division of labour (a few ambrosia beetles). Social evolution in this group has been regarded to be mostly driven by management of ectosymbiotic microbial communities (i.e., fungus farming), but whether this is indeed the case has to be shown. Also, it is assumed from other insect systems that the evolution of sociality is tightly linked with a host’s microbiome. For example, sociality will likely influence the mode of transgenerational microbiome transmission, leading to functional co-evolution between the host and its symbionts but also vice versa the symbionts may drive social evolution. Having differently social beetle systems provides us the option to study and understand potential “social” roles of symbionts in the systems.
People involved in this project:
Vienna Kowallik
Study systems:
Range of different bark and ambrosia beetles for comparable studies
Microbiome Evolution
Organisms adapt to their environment which is particularly important for bark beetles as they need to overcome the tree-defense mechanisms for survival. Microbiomes can be seen as a more flexible “extension” of the host genetic repertoire. In general, the capacity of the microbial community to change its composition, genetics or gene-expression pattern in response to selective forces can facilitate host rapid acclimation and adaptation to environmental change. This may play a role in the invasive success of some bark beetles or during population outbreaks of some bark beetle species, when they switch from preferring dead to living tree hosts. Therefore, we want to experimentally study if and how microbiomes evolve over several generations under certain selection factors and understand how these changes functionally affect the host.
People involved in this project:
Vienna Kowallik
Study systems:
In particular but not limited to: Xyleborinus saxesenii, Xylosandrus germanus, Ips typographus
